Autism is an Acquired Cellular Detoxification Deficiency Syndrome with Heterogeneous Genetic Predisposition
Volume 8 • Issue 1 • 1000224
Autism Open Access, an open access journal
ISSN: 2165-7890
DOI: 10.4172/2165-7890.1000224
James Lyons-Weiler*
Institute for Pure and Applied Knowledge, USA
Abstract
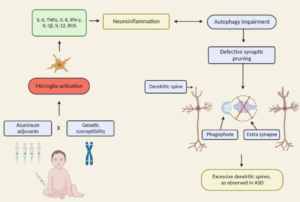
Neurodevelopmental disorders, including autism spectrum disorders, have a complex biological and medical basis involving diverse genetic risk and myriad environmental exposures. Teasing apart the role of specific stressors is made challenging due to the large number of apparently contributing associations, gene x environment interactions and phenomimicry. Historically, these conditions have been rare, making causality assessment at the population level infeasible. Only a few vaccines have been tested for association with autism, and it has been shown that improved diagnosis only explains a percentage of the increase in diagnosis. Now the rates are so high in some countries that public school programs cannot handle to large numbers of special needs students, and professionals are quitting their jobs due to security concerns. Here, I present a mechanistic biomedical process model (theory) of the pathophysiology of autism that reconciles the apparent paradox between the high degree of causal heterogeneity in environmental toxins, the absence of common “autism genes” and the high degree of genetic concordance (heritability) of ASD and ASD-like traits. In brief, the environmental toxin sampling liability for ASD varies among families involving different local exposures following injury to normal cellular endoplasmic detoxification and mitochondrial processes from toxic metals. The literature strongly supports that autism is most accurately seen as an acquired cellular detoxification deficiency syndrome with heterogeneous genetic predisposition that manifests pathophysiologic consequences of accumulated, run-away cellular toxicity. At a more general level, it is a form of a toxicant-induced loss of tolerance of toxins, and of chronic and sustained ER overload (“ER hyperstress”), contributing to neuronal and glial apoptosis via the unfolded protein response (UPR). Inherited risk of impaired cellular detoxification and circulating metal re-toxification in neurons and glial cells accompanied by chronic UPR is key. This model explains the aberrant protein disorder observed in ASD; the great diversity of genes that are found to have low, but real contributions to ASD risk and the sensitivity of individuals with ASD to environmental toxins. The hindrance of detoxification and loss of cellular energetics leads to apoptosis, release of cytokines and chronic neuroinflammation and microglial activation, all observed hallmarks of ASD. Interference with the development of normal complex (redundant) synapses leads to a pathological variation in neuronal differentiation, axon and dendrite outgrowth, and synaptic protein expression. The most general outcomes are overall simplification of gross synaptic anatomy and, neurofunctionally, a loss of inhibitory feedback and aberrations in long-term connections between distant regions of the brain. Failed resolution of the ER stress response leads to re-distribution of neurotoxic metals, and the impaired neurocellular processes lead to subsequent accumulation of a variety of additional types of toxins with secondary, sometime life-threatening comorbidities such as seizures, with overlapping (not mutually exclusive) causality. Reduction of exposure to toxins known to cause mitopathy (mercury) and endoplasmic reticulum dysfunction (mercury and aluminum) during pregnancy and during the early years of development will reduce the risk of ER overload and ER hyperstress, and of ASD diagnosis. This knowledge has immediate clinical translational relevance: Post-vaccination symptoms should be heeded as a sign of susceptibility to toxin; Vitamin D can be increased to drive the healthy early phases of the unfolded protein response (UPR), and mutations in ASD genes encoding proteins with high intrinsic disorder may contraindicate the use of aluminum and mercury for carriers of risk alleles. Clinicians should be alert to a patient’s Vitamin D receptor (BSM) mutational status prior to recommending increased doses. Approaches to improving overall brain health in autistics must be de-stigmatized and given high priority. Reduction of lifetime exposures of industrial and agricultural toxins will improve brain health for the entire human population. Purely genetic studies of ASD, and studies that do not include vaccination as an environmental exposure with potential liability and interactions with genes, are unethical. To qualify as science, studies must test plausible hypotheses, and the absence of association from poorly designed, unethically executed, and underpowered and unsound whole-population association studies have been harmful distractions in the quest for understanding. Skilled pediatricians and ob/gyns will seek evidence of genetic predisposition to environmental susceptibility in the form of non-synonymous substitutions in brain proteins that require ER-folding, and they will provide informed cautions on exposures (from all sources) to environmental toxins to patients and parents of patients with signs of metal and chemical sensitivity. To aid in this, a list of ASD environmental susceptibility protein-encoded genes is presented. A clinical exome sequence test, followed by loss-of-function prediction analysis, would point to individuals most susceptible to vaccine metal-induced ER hyper stress.