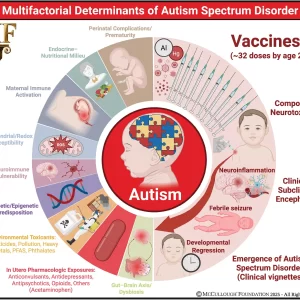
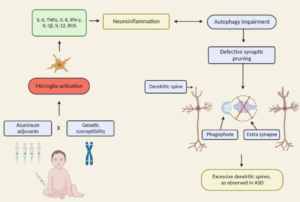
Conclusion: The totality of evidence supports a multifactorial model of ASD in which genetic predisposition, neuroimmune biology, environmental toxicants, perinatal stressors, and iatrogenic exposures converge to produce the phenotype of a post-encephalitic state. Combination and early-timed routine childhood vaccination constitutes the most significant modifiable risk factor for ASD, supported by convergent mechanistic, clinical, and epidemiologic findings, and characterized by intensified use, the clustering of multiple doses during critical neurodevelopmental windows, and the lack of research on the cumulative safety of the full pediatric schedule. As ASD prevalence continues to rise at an unprecedented pace, clarifying the risks associated with cumulative vaccine dosing and timing remains an urgent public health priority.
Mitochondria
Small structures in a cell that are found in the cytoplasm (fluid that surrounds the cell nucleus). Mitochondria make most of the energy for the cell and have their own genetic material that is different from the genetic material found in the nucleus. Many diseases are caused by mutations (changes) in the DNA of mitochondria. Mitochondria are cell organelles. – NCI Dictionary of Cancer Terms, U.S. National Cancer Institute, 2021
Exerpt
“any adjuvant or condition boosting immune activation of certain cells, such as Central Nervous System (CNS) microglia and macrophages, if sequentially activated by the vaccination process, whatever the adjuvant or immune stimulation principle, will trigger this mechanism, not only in the CNS, but possibly other peripheral organs and tissues containing glutamate receptors”
Highlights
• Thimerosal (TM) significantly affects mitochondrial bioenergetics in the brain.
• Mitochondrial integrity (membrane potential) was maintained after acute TM treatment.
• Ethylmercury released after the breakdown of TM compromised the cholinergic system.
• The brain is more sensitive to oxidative stress induced by TM compared to the liver.
Excerpt:
“Our findings of significantly altered acylcarnitine levels in ASD support the hypothesis of altered fatty acid metabolism in a subset of ASD patients.”
Press Release from Harvard Magazine:
“Inflammation link for autism
A neuroimaging study has shown that the brains of young men with autism spectrum disorder have low levels of translocator protein, a substance that appears to play a role in inflammation and metabolism.
This discovery by a team of HMS researchers at Massachusetts General Hospital provides an important insight into the possible origins of autism spectrum disorder.
This developmental disorder, which affects one in fifty-nine children in the United States, emerges in early childhood and is characterized by difficulty communicating and interacting with others. Although the cause is unknown, growing evidence has linked it to neuroinflammation.
One sign of neuroinflammation is elevated levels of translocator protein, which can be measured in the brain using positron-emission tomography and anatomic magnetic resonance imaging.
The research team used these imaging tools to scan the brains of fifteen young adult males with the disorder. The group included both high- and low-functioning participants with varying degrees of intellectual ability. As a control, the team scanned the brains of eighteen non-autistic young men of similar age.
The scans showed that the brains of the young men with the disorder had lower levels of the protein, compared with the brains of non-autistic participants. In fact, those participants with the most severe symptoms of the disorder tended to have the lowest expression of the protein.
The brain regions found to have low expression of the protein have previously been linked to autism spectrum disorder and are thought to govern social and cognitive capacities such as processing emotions, interpreting facial expressions, and empathy.
The researchers point out that the translocator protein has multiple complex roles, some of which promote brain health. Adequate levels of the protein are, for example, necessary for normal functioning of mitochondria. Earlier research has linked malfunctioning mitochondria in brain cells to autism spectrum disorder.
Zürcher NR et al., Molecular Psychiatry, February 2020
Excerpt:
“The literature strongly supports that autism is most accurately seen as an acquired cellular detoxification deficiency syndrome with heterogeneous genetic predisposition that manifests pathophysiologic consequences of accumulated, run-away cellular toxicity. At a more general level, it is a form of a toxicant-induced loss of tolerance of toxins, and of chronic and sustained ER overload (“ER hyperstress”), contributing to neuronal and glial apoptosis via the unfolded protein response (UPR). Inherited risk of impaired cellular detoxification and circulating metal re-toxification in neurons and glial cells accompanied by chronic UPR is key.”
Abstract
The conceptualisation of autistic spectrum disorder and Alzheimer’s disease has undergone something of a paradigm shift in recent years and rather than being viewed as single illnesses with a unitary pathogenesis and pathophysiology they are increasingly considered to be heterogeneous syndromes with a complex multifactorial aetiopathogenesis, involving a highly complex and diverse combination of genetic, epigenetic and environmental factors. One such environmental factor implicated as a potential cause in both syndromes is aluminium, as an element or as part of a salt, received, for example, in oral form or as an adjuvant. Such administration has the potential to induce pathology via several routes such as provoking dysfunction and/or activation of glial cells which play an indispensable role in the regulation of central nervous system homeostasis and neurodevelopment. Other routes include the generation of oxidative stress, depletion of reduced glutathione, direct and indirect reductions in mitochondrial performance and integrity, and increasing the production of proinflammatory cytokines in both the brain and peripherally. The mechanisms whereby environmental aluminium could contribute to the development of the highly specific pattern of neuropathology seen in Alzheimer’s disease are described. Also detailed are several mechanisms whereby significant quantities of aluminium introduced via immunisation could produce chronic neuropathology in genetically susceptible children. Accordingly, it is recommended that the use of aluminium salts in immunisations should be discontinued and that adults should take steps to minimise their exposure to environmental aluminium.
Excerpt:
“Environmental mercury is neurotoxic at doses well below the current reference levels considered to be safe, with evidence of neurotoxicity in children exposed to environmental sources including fish consumption and ethylmercury-containing vaccines. Possible neurotoxic mechanisms of mercury include direct effects on sulfhydryl groups, pericytes and cerebral endothelial cells, accumulation within astrocytes, microglial activation, induction of chronic oxidative stress, activation of immune-inflammatory pathways and impairment of mitochondrial functioning. (Epi-)genetic factors which may increase susceptibility to the toxic effects of mercury in ASD include the following: a greater propensity of males to the long-term neurotoxic effects of postnatal exposure and genetic polymorphisms in glutathione transferases and other glutathione-related genes and in selenoproteins. Furthermore, immune and inflammatory responses to immunisations with mercury-containing adjuvants are strongly influenced by polymorphisms in the human leukocyte antigen (HLA) region and by genes encoding effector proteins such as cytokines and pattern recognition receptors. Some epidemiological studies investigating a possible relationship between high environmental exposure to methylmercury and impaired neurodevelopment have reported a positive dose-dependent effect.”
Conclusion The literature reviewed here suggests a link between abnormalities in mitochondrial homeostasis and ASD and provides biochemical and genetic evidence to support a role for mitochondrial dysfunction in the pathogenesis of the autism phenotype.
Excerpt:
“Children who have (mitochondrial-related) dysfunctional cellular energy metabolism might be more prone to undergo autistic regression between 18 and 30 months of age if they also have infections or immunizations at the same time.”