Abstract
In July 2025, Andersson et al. reported in Annals of Internal Medicine that early-life exposure to Aluminium (Al)-adjuvanted vaccines was not associated with increased risk of 50 chronic diseases, based on a Danish cohort of 1.2 million children. While widely cited as reassuring evidence of Al-Based Adjuvant (ABA) safety, closer scrutiny reveals major methodological and conceptual flaws. Specifically, the study demonstrates limited understanding of Al toxicology, weaknesses in cohort design and statistical analysis, and insufficient transparency regarding potential conflicts of interest. We argue that these shortcomings prevent meaningful conclusions about ABA safety, particularly in relation to neurodevelopmental and autoimmune outcomes, and highlight the need for more rigorous, transparent, and scientifically grounded investigations.
February 2026
Aluminum / Blood brain barrier / Bradford Hill criteria / Epidemiology / HLA / IL-1β / Lymphocytes / MTHFR / Neuroinflammation / NLRP3 / Toxicants
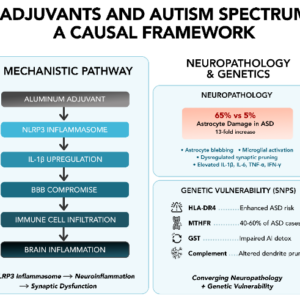
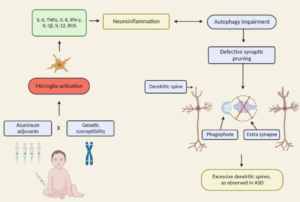
Conclusions: Converging mechanistic, neuropathological, epidemiological, and genetic
evidence demonstrates that aluminum adjuvants can trigger ASD in genetically susceptible
individuals through well-characterized neuroinflammatory pathways. The 80-fold increase
in ASD prevalence temporally correlating with vaccine schedule expansion, combined with
robust biological mechanisms and postmortem findings, demands urgent re-examination of aluminum adjuvant safety in the context of neurodevelopment, particularly in genetically vulnerable populations.