Conclusions: Converging mechanistic, neuropathological, epidemiological, and genetic evidence demonstrates that aluminum adjuvants can trigger ASD in genetically susceptible individuals through well-characterized neuroinflammatory pathways. The 80-fold increase in ASD prevalence temporally correlating with vaccine schedule expansion, combined with robust biological mechanisms and postmortem findings, demands urgent re-examination of aluminum adjuvant safety in the context of neurodevelopment, particularly in genetically vulnerable populations.
Blood brain barrier
A network of blood vessels and tissue that is made up of closely spaced cells and helps keep harmful substances from reaching the brain. The blood-brain barrier lets some substances, such as water, oxygen, carbon dioxide, and general anesthetics, pass into the brain. It also keeps out bacteria and other substances, such as many anticancer drugs.
NCI Dictionary of Cancer Terms
U.S. National Cancer Institute, 2021
Excerpts:
“As a result of these pieces of evidence (epidemiological, clinical and preclinical data) pointing to a potential causal association between early ABA (aluminum-based adjuvants) exposure and increased ASD risk, new hypotheses regarding neurological and immunological consequences of ABA-containing vaccines and novel clinical strategies (i.e., postponing of ABA-containing vaccines and replacement of ABAs with calcium phosphate are now being considered.“
“Our review presents the lack of fundamental scientific data demonstrating that Al adjuvants are safe and do not induce any long-term side effects. It also supports further investigation related to the effects of early Al adjuvant exposures occurring in combination with genetic susceptibility factors, including autophagy, immune and inflammation process genes. As accumulating evidence shows that modulating the levels of autophagy may increase the risk of NDDs, such studies will elucidate a new etiology for these complex disorders and contribute to develop potential new diagnostic and therapeutic tools.”
Excerpt:
“This finding suggests that clinical events concerning neonatal IL-4 over-exposure, including neonatal hepatitis B vaccination and allergic asthma in human infants, may have adverse implications for brain development and cognition.”
Excerpts:
“Herein, we will discuss the accumulating literature for ASD, giving special attention to the relevant aspects of factors that may be related to the neuroimmune interface in the development of ASD, including changes in neuroplasticity.”
Commentary on the article:
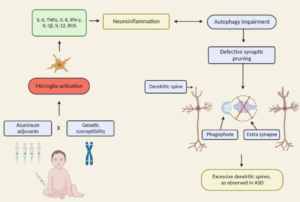
“The authors rightly highlight the newest challenging frontier of autism research: the neuroimmune axis alterations. These alterations are first evident in the cells early responsible for immune responses, as they are the precursors for macrophages, dendritic, and microglial cells: monocytes or peripheral blood mononuclear cells (PBMCs). These cells show strong dysfunctions in ASD children and are committed to a pro-inflammatory state, which in turn result in long-term immune alterations (4). In ASDs, altered PBMCs are responsible for elevated pro-inflammatory cytokine production. The up-regulation of inflammatory cytokines is also reflected in brain centers of autistic patients (5): the consequences are the induction of blood–brain barrier (the immunological interface between peripheral immune system and central nervous system) disruption. Changes in BBB permeability directly influence neural plasticity, connectivity and function, triggering impairments in social interaction, communication, and behavior (3). Immunological abnormalities also influence the gastrointestinal system and the microglial innate immune cells of the central nervous system (6). The authors also discuss the role of autoimmunity in the pathogenesis of autism. Familial or virus/bacteria-infected autoimmunity could be a risk factor for autism. Even if the exact cellular and molecular pathways responsible for the induction of neuroimmune alterations are still to be further clarify, a complex interaction among epigenetic and environmental risk factors (7) could trigger the neuroimmune abnormalities, such as abnormal neuron and glia responses.”
Conclusions
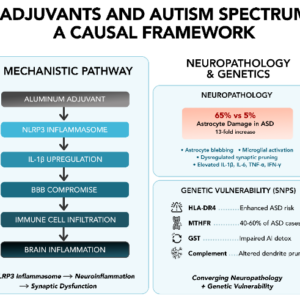
In the ASD brain, there is an altered expression of genes associated with BBB integrity coupled with increased neuroinflammation and possibly impaired gut barrier integrity.
Excerpt
“Male mice in the “high Al” group showed significant changes in light–dark box tests and in various measures of behaviour in an open field. Female mice showed significant changes in the light–dark box at both doses, but no significant changes in open field behaviours. These current data implicate Al injected in early postnatal life in some CNS alterations that may be relevant for a better understanding of the aetiology of ASD.”
Excerpts:
“Intramuscular injection of alum-containing vaccine was associated with the appearance of aluminum deposits in distant organs, such as spleen and brain where they were still detected one year after injection.”
“Cerebral translocation was not observed after direct intravenous injection, but significantly increased in mice with chronically altered blood-brain-barrier.”
Conclusion
Nanomaterials can be transported by monocyte-lineage cells to DLNs, blood and spleen, and, similarly to HIV, may use CCL2-dependent mechanisms to penetrate the brain. This occurs at a very low rate in normal conditions explaining good overall tolerance of alum despite its strong neurotoxic potential. However, continuously escalating doses of this poorly biodegradable adjuvant in the population may become insidiously unsafe, especially in the case of overimmunization or immature/altered blood brain barrier or high constitutive CCL-2 production.
Excerpt:
“Therefore, autism is the result of genetic defects and/or inflammation of the brain. The inflammation could be caused by a defective placenta, immature blood-brain barrier, the immune response of the mother to infection while pregnant, a premature birth, encephalitis in the child after birth, or a toxic environment.”
Conclusions: HgCl2 stimulates VEGF and IL-6 release from human mast cells. This phenomenon could disrupt the blood-brain-barrier and permit brain inflammation. As a result, the findings of the present study provide a biological mechanism for how low levels of mercury may contribute to ASD pathogenesis.
Excerpt:
“There is significant evidence that the pathogenesis of several neurodegenerative diseases, including Parkinson’s disease, Alzheimer’s disease, Friedreich’s ataxia and amyotrophic lateral sclerosis, may involve the generation of reactive oxygen species and mitochondrial dysfunction.”