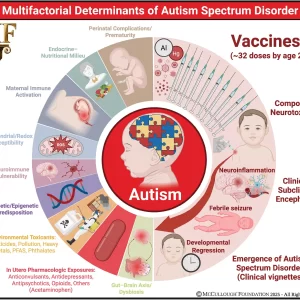
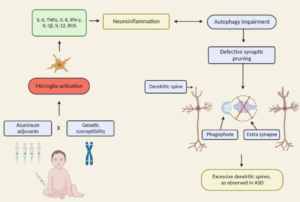
Excerpts:
“The likelihood of the child having ASD more than doubled among children with food allergy compared with those without food allergy; children with respiratory and skin allergy were also significantly more likely to have ASD, but at a lesser magnitude. While no sex difference was found for food allergy, boys with ASD were significantly more likely than girls with ASD to have respiratory and skin allergy.”
” It may be that GI dysfunction, seizures, and sleep disorder, in addition to food, respiratory, and skin allergies, are medical comorbidities that characterize the immune-mediated subtype of ASD.”
“In the Discussion section of their article, Xu and colleagues review other aspects of immune dysfunction reported in ASD, including abnormalities in peripheral immunoglobulins, imbalance of T-cell subsets, and increased levels of proinflammatory cytokines in postmortem brains of patients with ASD. Considering the significant association between food, respiratory, and skin allergy in children with ASD reported by Xu and colleagues, in conjunction with numerous studies documenting aspects of immune dysfunction in patients with ASD and specific animal models of ASD, evidence continues to mount that an immune-mediated subtype of ASD should continue to be pursued and defined.”
More